Application of rhenium metals in catalysts
- Categories:Application Technology
- Author:Rheniumet
- Origin:
- Time of issue:2022-11-29
- Views:0
(Summary description)Rhenium (Re) and its compounds have excellent catalytic activity (Re has a highly selective catalytic function for many chemical reactions), so it is often used as a catalyst in petrochemical and other fields. Catalytic reforming is one of the petroleum refining processes and an important means to improve the quality of gasoline and produce petrochemical raw materials. As a catalyst in catalytic reforming process, Re - Pt alloy can increase the depth of reforming reaction and increase the yield of gasoline, aromatic hydrocarbons and hydrogen. In addition, rhenium can also be used as a catalyst for automobile exhaust purification, and rhenium sulfide can be used as a hydrogenation catalyst for cresol, lignin, etc. The following lists the application of several rhenium metal catalysts.
Application of rhenium metals in catalysts
(Summary description)Rhenium (Re) and its compounds have excellent catalytic activity (Re has a highly selective catalytic function for many chemical reactions), so it is often used as a catalyst in petrochemical and other fields. Catalytic reforming is one of the petroleum refining processes and an important means to improve the quality of gasoline and produce petrochemical raw materials. As a catalyst in catalytic reforming process, Re - Pt alloy can increase the depth of reforming reaction and increase the yield of gasoline, aromatic hydrocarbons and hydrogen. In addition, rhenium can also be used as a catalyst for automobile exhaust purification, and rhenium sulfide can be used as a hydrogenation catalyst for cresol, lignin, etc. The following lists the application of several rhenium metal catalysts.
- Categories:Application Technology
- Author:Rheniumet
- Origin:
- Time of issue:2022-11-29
- Views:0
Rhenium (Re) and its compounds have excellent catalytic activity (Re has a highly selective catalytic function for many chemical reactions), so it is often used as a catalyst in petrochemical and other fields. Catalytic reforming is one of the petroleum refining processes and an important means to improve the quality of gasoline and produce petrochemical raw materials. As a catalyst in catalytic reforming process, Re - Pt alloy can increase the depth of reforming reaction and increase the yield of gasoline, aromatic hydrocarbons and hydrogen. In addition, rhenium can also be used as a catalyst for automobile exhaust purification, and rhenium sulfide can be used as a hydrogenation catalyst for cresol, lignin, etc. The following lists the application of several rhenium metal catalysts.

1. supported type of Re - based catalyst C, alkene disproportionation to propylene
Supported Re based catalysts are mainly composed of support, active components, and sometimes other alkali metals, alkaline earth metals and other additives are added to improve the performance of the catalyst. "Carriers generally use inorganic oxides with high specific surface area (such as Al203, SiO2, B2O3, Fe203, etc.) or their composite oxides (such as SiO2/Al203, B2O3/SiO2/Al203, Fe203/Al203, Al203-B2O3, etc.)." The active components are oxides (such as Re203, Re02, Re03 and Re207) or their precursors ammonium perrhenate, perrhenate acid, etc.; The preparation method of the catalyst is generally impregnated, the carrier is impregnated in a certain concentration of rhenium metal brine solution, and then dried, and calcined at high temperature in the air flow. During the calcination process, the salts are decomposed to obtain the precursor of the supported rhenium based catalyst, which is then pretreated to obtain the supported rhenium based catalyst. Due to the high selectivity and activity at low temperature, negative rheni-based catalysts have attracted wide attention of researchers. Supported rheni-based catalysts have been used in the alkene dismutation process developed by BASF in Germany, and the Meta-4-ene dismutation process developed by IFP in France and China National Petroleum Corporation in Taiwan.
2. Hydrogenation of CO2 to methanol catalyzed by indium oxide supported rhenium catalyst
With the continuous development of renewable hydrogen energy technology, the selective hydrogenation of CO2 to methanol has been widely concerned around the world. How to prepare catalysts with high activity, high selectivity and high stability is very important. The results show that In2O3 with oxygen vacancy has high selectivity for the synthesis of methanol by hydrogenation of CO2. The calculation shows that the surface of In2O3 under the action of hydrogen generates oxygen vacancy, CO2 is activated on the oxygen vacancy, the activated CO2 undergoes hydrogenation reaction to methanol desorption, the vacancy disappears, and the In2O3 is restored to its normal state, forming a perfect cycle. Due to the high selectivity of In2O3 catalyst for methanol and the clear active site, related research works soon attracted extensive attention from domestic and foreign counterparts. In order to improve the hydrogenation activity of indium oxide catalyst, Mei Donghai and Liu Changjun from Tianjin University of Technology loaded metal rhenium on indium oxide and controlled the size of Re catalyst by adjusting Re loading, so that it was highly dispersed on In2O3 carrier and had strong metal-carrier interaction with In2O3. Furthermore, the catalytic activity and stability of indium oxide CO2 hydrogenation to methanol were improved. It provides a new idea for understanding the structure-property relationship of In2O3-based catalysts and developing new efficient CO2 hydrogenation catalysts.
3, high Re ultra low Pt CB-8 reforming catalyst
Rhenium is an excellent catalyst for hydrogenation and isomerization, which can be used for catalytic reforming of naphtha in gasoline (rhenium process). Platinum rhenium catalysts are mainly used in the manufacture of lead-free, high octane gasoline. The high rhenium and low Pt CB-8 catalyst with 0.15w Pt content is a new generation of industrial reforming catalyst developed by Fushun Petrochemical Research Institute. The industrial application began in 1991. The results showed that the average inlet temperature was 495C, the aromatic hydrocarbon conversion rate was 125w %, the activity loss was about 2C after 317 days of operation, and the carbon accumulation was only 5.55w %, which completely reached or exceeded the results of the medium-sized test. The reaction performance of the CB-8 catalyst recovered to the level of the new catalyst after regenerating chlorination, indicating that the CB-8 catalyst was a new type of reforming catalyst with excellent activity, selectivity, stability, regeneration performance and mechanical strength. Although PT - Re reforming catalyst has been industrialized for a long time, its performance is still being improved, and its mechanism is still being studied.

4.Rhenium (molybdenum) functional compounds catalyze cellulose hydrolysis
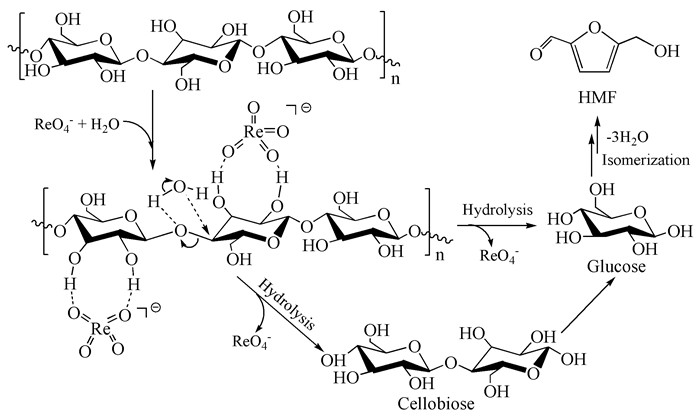
The transition metal rhenium and molybdenum compounds also have some catalytic effects on the hydrolysis of carbohydrates. Some research groups have also done a lot of work on the hydrolysis and transformation of cellulose, and have successfully applied the functional compounds methyl rhenium trioxide (MTO)[59] and perrhenate [60] to the hydrolysis reaction of cellulose. Among them, tetramethylammonium perrhenate had the most significant catalytic activity on microcrystalline cellulose. The yield of sugar and glucose was 97.1% and 42%, respectively, when microcrystalline cellulose was heated at 150℃ for 30min with microwave assisted ionic liquid as solvent. As for the reaction mechanism, Re=O and cellulose are dissolved in ionic liquid and in full contact with each other. Re=O forms a strong hydrogen bond with the hydroxyl group of cellulose molecules, which makes the β-1, 4-glycosidic bond in cellulose molecules weak or even broken, and promotes the hydrolysis of cellulose to small molecular sugars such as glucose and cellobiose.
Scan the QR code to read on your phone
Contact Us
Address: Room 401, Building 10, Xinggong Science and Technology Park, No. 100 Luyun Road, Changsha High-tech Development Zone, Hunan
Email:sales@rheniumet.com

scan it Follow us
Copyright: Hunan Rhenium Alloy Material Co., Ltd. ICP No. 13005464-1 Website construction: www.300.cn



